Introduction to Medicine Field Risks in Singapore
The medicine field in Singapore, a global hub for advanced healthcare, operates within a uniquely dynamic environment. Understanding its inherent risks is critical for patient safety, fostering innovation, and maintaining the nation’s healthcare system integrity. This introduction defines the scope and importance of comprehending challenges faced by pharmaceutical stakeholders. It underscores the necessity of proactively identifying, assessing, and mitigating risks, especially with technological advancements, evolving global supply chains, and emerging health threats. Insights from the trend report of the risk of medicine field in singapore are indispensable for strategic planning and operational resilience in this high-stakes domain.

-
Defining ‘Risk’ in Singapore’s Pharmaceutical Landscape
In Singapore’s sophisticated pharmaceutical landscape, ‘risk’ encompasses potential events adversely impacting medicinal product quality, safety, efficacy, availability, or financial viability. These extend beyond traditional business hazards into critical public health areas. Regulatory risks involve non-compliance with rigorous Health Sciences Authority (HSA) standards, potentially leading to recalls. Supply chain vulnerabilities pose significant threats, given Singapore’s reliance on global sources for active pharmaceutical ingredients (APIs) and finished products. Geopolitical instability, natural disasters, or pandemics can disrupt these chains, causing drug shortages affecting patient outcomes.
Other risks include market competition, patent expirations, and operational failures like manufacturing defects or human error. Cybersecurity risks threaten sensitive patient data and intellectual property. Patient safety introduces risks related to adverse drug reactions and medication errors. Robust, tailored risk management frameworks are crucial. Understanding these multifaceted dimensions is foundational for effective mitigation strategies.
-
Why a Trend Report for 2026 is Crucial for Stakeholders
A comprehensive trend report on risks within Singapore’s medicine field for 2026 is a forward-looking strategic tool. Manufacturers and distributors gain crucial foresight into emerging regulatory changes, potential supply chain disruptions, and shifting market dynamics, enabling adaptive business models. Healthcare providers leverage this intelligence to anticipate drug shortages and enhance patient safety protocols.
Regulators like the HSA identify areas needing strengthened frameworks or new guidelines for nascent risks from advanced therapies and digital health. Investors gain a clearer picture of industry stability. Ultimately, patients are the primary beneficiaries, as proactive risk management, informed by such reports, translates into safer, more effective, and reliably available medicines. In an industry of rapid innovation and unpredictable global events, a detailed trend report of the risk of medicine field in singapore provides the strategic compass needed to navigate complexity, fostering resilience and sustainable growth.
-
Overview of Singapore’s Healthcare Ecosystem and Regulations
Singapore boasts one of the most efficient and high-performing healthcare systems globally, characterized by universal access and high-quality care. This robust ecosystem is anchored by world-class medical facilities and a skilled workforce. Central to ensuring medicinal product safety and efficacy is a sophisticated regulatory framework. The Health Sciences Authority (HSA) serves as the primary regulatory body, responsible for licensing, quality control, and post-market surveillance of therapeutic products, medical devices, and health products. HSA’s oversight spans a medicine’s entire lifecycle, from clinical trials to pharmacovigilance.
Singapore’s regulatory approach aligns with international standards, ensuring products meet global benchmarks for quality, safety, and efficacy. The Ministry of Health (MOH) plays a pivotal role in policy formulation and strategic planning. This stringent regulatory environment, while demanding, builds public trust and safeguards national health. For more on Singapore’s health product regulatory landscape, refer to the official Health Products Regulations section on the HSA website. This comprehensive oversight forms the bedrock for all risk assessments and mitigation strategies in the medicine field.
Key Trends Influencing Medicine Risks (2023-2026)
Singapore’s pharmaceutical landscape, a pivotal hub in Asia, is navigating a complex web of macro and micro trends that profoundly shape the risk profile of medicine. From rapidly evolving regulatory frameworks to groundbreaking technological advancements and the persistent specter of global events, understanding these dynamics is crucial for stakeholders across the entire value chain. This section delves into the trend report of the risk of medicine field in singapore, highlighting key areas where risks are escalating and mitigation strategies are becoming increasingly vital. The coming years, 2023-2026, promise a period of significant transformation, demanding agility and foresight from pharmaceutical companies, healthcare providers, and regulatory bodies alike.
1. Regulatory Evolution and Compliance Challenges in Pharma
The regulatory environment governing pharmaceuticals in Singapore and globally is in a constant state of flux, driving both innovation and significant compliance challenges. The Health Sciences Authority (HSA) continues to refine its frameworks, often aligning with international best practices from bodies like ICH (International Council for Harmonisation) and PIC/S (Pharmaceutical Inspection Co-operation Scheme). This evolution is marked by heightened scrutiny over product quality, safety, and efficacy, impacting everything from drug development and clinical trials to manufacturing, distribution, and post-market surveillance. New guidelines for novel therapies, such as cell and gene therapies, and digital health products, necessitate sophisticated compliance strategies. Data integrity remains a paramount concern, with regulators demanding robust systems to prevent fraud and ensure the reliability of all submitted data. Companies face increasing pressure to invest in advanced quality management systems, comprehensive training, and cutting-edge technology to meet these evolving standards. The cost of non-compliance, ranging from hefty fines and product recalls to reputational damage, underscores the critical importance of proactive engagement with these regulatory shifts. Staying abreast of both local mandates and international harmonization efforts is not merely a legal obligation but a strategic imperative for any entity operating within the medicine risk landscape in Singapore.
2. Impact of Digitalization, AI, and Telemedicine on Safety
The rapid integration of digitalization, Artificial intelligence (AI), and telemedicine is fundamentally reshaping the healthcare ecosystem, offering unprecedented opportunities for efficiency and patient care while simultaneously introducing new layers of risk in the medicine field. AI-driven solutions are revolutionizing drug discovery, personalized medicine, and pharmacovigilance, enabling faster analysis of vast datasets to identify adverse drug reactions and predict patient responses more accurately. Telemedicine platforms have expanded access to healthcare, particularly vital during public health crises, allowing for remote consultations, prescription renewals, and monitoring. However, these advancements come with inherent risks. Data security and patient privacy are significant concerns, as the volume of sensitive health information shared digitally grows exponentially, making systems vulnerable to cyber threats. The accuracy and ethical implications of AI algorithms are also under scrutiny; algorithmic bias can lead to misdiagnoses or inappropriate treatment recommendations, particularly in diverse patient populations. Moreover, the regulatory landscape for Software as a Medical Device (SaMD) is still maturing, posing challenges in ensuring the safety and efficacy of AI-powered diagnostic tools and digital therapeutics. The potential for miscommunication or diagnostic errors in remote consultations also needs careful management. Mitigating these risks requires robust cybersecurity measures, transparent AI development, continuous training for healthcare professionals, and clear regulatory guidelines that keep pace with technological innovation, ensuring that the benefits of digitalization do not inadvertently compromise patient safety.
3. Emerging Pathogens and Global Supply Chain Vulnerabilities
The COVID-19 pandemic served as a stark reminder of the profound impact emerging pathogens can have on global health and the fragility of medicine supply chains. The rise of new viral strains and the growing threat of antimicrobial resistance (AMR) continue to pose significant risks, potentially triggering new public health emergencies that disrupt the availability and distribution of essential medicines. Geopolitical tensions, trade disputes, and natural disasters further exacerbate these vulnerabilities, leading to shortages of critical active pharmaceutical ingredients (APIs), excipients, and finished products. Many pharmaceutical supply chains are long and complex, relying heavily on single-source suppliers or specific geographic regions, making them susceptible to localized disruptions. The race for vaccines and therapeutics during recent crises highlighted the fierce competition for raw materials and manufacturing capacity, often leaving smaller nations or those with less purchasing power at a disadvantage. To build resilience, the industry is increasingly focused on diversification of suppliers, regionalization of manufacturing, and the implementation of advanced supply chain visibility tools. Governments and pharmaceutical companies are exploring strategies like strategic stockpiling, local manufacturing incentives, and international collaborations to ensure a more robust and responsive supply of medicines. Addressing these vulnerabilities is paramount to safeguarding public health and ensuring access to life-saving treatments, especially as the world anticipates future health crises. The World Health Organization (WHO) has consistently highlighted the urgency of global action against threats like AMR and supply chain instability, underscoring the interconnected nature of these risks.
Major Risk Categories Identified in the Medicine Field
The medicine field, a cornerstone of public health, is perpetually exposed to a complex array of risks that evolve with scientific advancements, global interconnectivity, and digital transformation. In a sophisticated healthcare hub like Singapore, understanding and mitigating these risks are paramount to safeguarding patient well-being, maintaining clinical excellence, and preserving the integrity of the healthcare system. A thorough examination of the trend report of the risk of medicine field in singapore reveals several critical categories that demand continuous vigilance from both providers and regulators, ensuring resilient and trusted medical services for all.
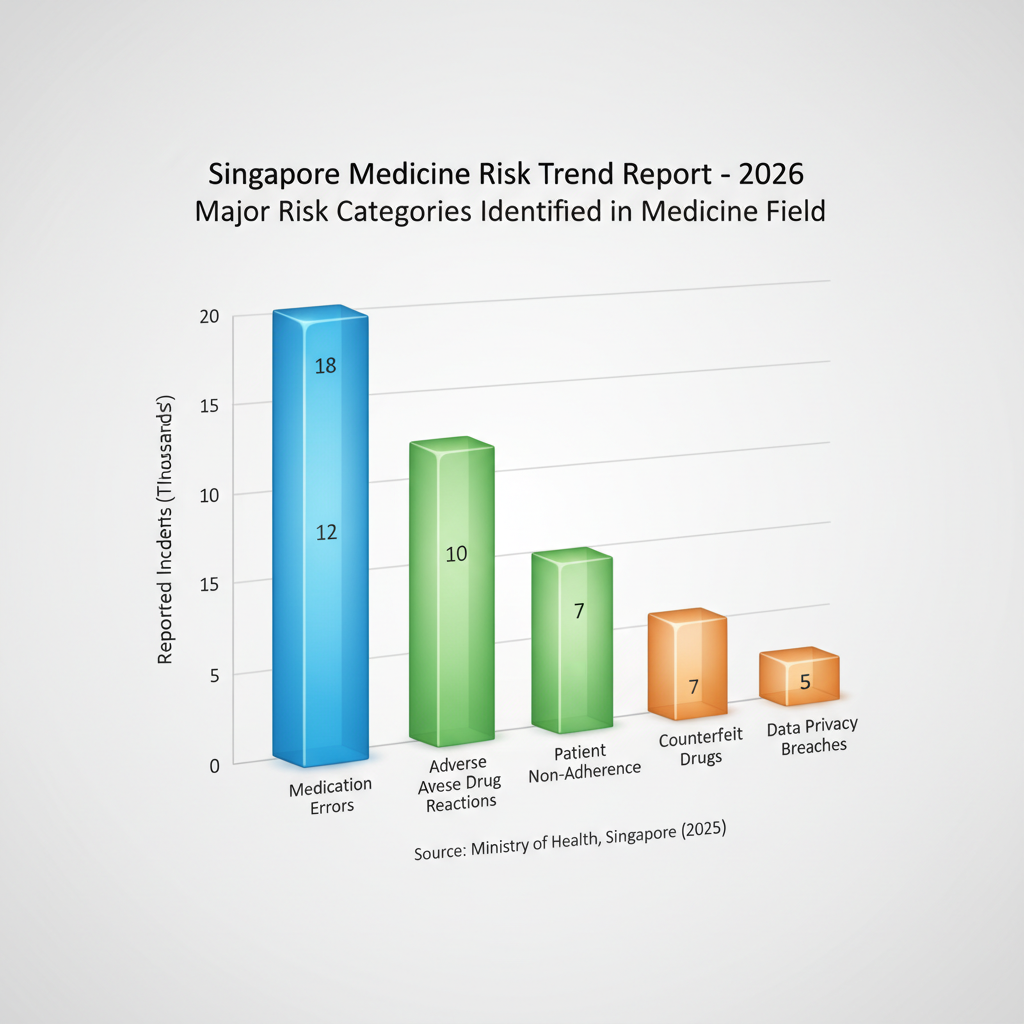
1. Pharmacovigilance and Adverse Drug Reaction Risks
Pharmacovigilance, the science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problem, stands as a critical pillar of patient safety. Despite rigorous clinical trials, not all potential side effects or drug interactions can be identified before a medicine reaches the market. Adverse Drug Reactions (ADRs) can range from mild, manageable discomforts to severe, life-threatening events, impacting patient quality of life and potentially increasing healthcare burdens.
In Singapore, the Health Sciences Authority (HSA) plays a pivotal role in post-market surveillance, collecting and analyzing ADR reports. However, the sheer volume of new drugs, combination therapies, and the diverse patient demographics present ongoing challenges. For instance, an unexpected severe ADR to a commonly prescribed medication, if not promptly identified and communicated, could lead to widespread patient harm, especially among vulnerable populations such as the elderly or those with multiple co-morbidities. Ensuring robust drug safety in Singapore requires continuous education for healthcare professionals on reporting, active patient engagement, and swift regulatory responses to emergent safety signals.
2. Counterfeit Medicines and Supply Chain Integrity Threats
The globalized pharmaceutical supply chain, while efficient, is unfortunately vulnerable to infiltration by counterfeit medicines. These illicit products, which may contain incorrect ingredients, no active ingredients, or even harmful substances, pose a direct threat to patient health, leading to treatment failures, drug resistance, and severe adverse outcomes. Singapore, as a significant trading hub with advanced logistics capabilities, is not immune to these threats, despite its stringent import controls.
The integrity of the pharmaceutical supply chain can be compromised at various stages, from manufacturing and packaging to distribution and dispensing. The rise of unregulated online pharmacies further complicates matters, making it easier for counterfeiters to reach unsuspecting patients. The economic impact of counterfeit drugs is also substantial, eroding trust in legitimate pharmaceutical companies and potentially funding organized crime. Protecting the pharmaceutical supply chain security is a complex endeavor that requires international collaboration, advanced authentication technologies, and robust enforcement to prevent these dangerous products from reaching patients in Singapore and beyond.
3. Data Privacy, Cybersecurity, and Intellectual Property Risks
The digital transformation of healthcare has brought immense benefits but also introduced significant risks related to data privacy, cybersecurity, and intellectual property (IP). Healthcare institutions in Singapore, like globally, manage vast amounts of highly sensitive patient data, including electronic health records (EHRs), genetic information, and diagnostic images. A breach of this data, whether through ransomware attacks, phishing, or insider threats, can lead to identity theft, financial fraud, medical complications, and a severe erosion of patient trust. Maintaining medical data security Singapore is paramount.
Beyond patient data, the intellectual property of pharmaceutical companies represents colossal investments in research and development. Singapore’s burgeoning biomedical sciences sector, home to numerous innovative drug manufacturers and research institutions, is a prime target for IP theft and corporate espionage. The loss of proprietary drug formulas, clinical trial data, or manufacturing processes can severely impact a company’s competitive advantage, stifle innovation, and ultimately delay the availability of new treatments. Robust cybersecurity measures, stringent data governance policies, and strong legal frameworks for IP protection are essential to safeguard both patient information and the future of medical innovation in Singapore.
Mitigation Strategies and Best Practices for Risk Management
Singapore’s medicine sector, a vital component of its thriving healthcare landscape, faces an intricate web of risks ranging from global supply chain disruptions and the rise of counterfeit medicines to evolving regulatory landscapes and the rapid pace of technological change. Proactive risk management is not merely a compliance exercise but a strategic imperative for ensuring patient safety, maintaining public trust, and safeguarding the nation’s health security. Moving beyond reactive measures, organizations in Singapore are increasingly adopting robust mitigation strategies and industry best practices. These efforts aim to not only address identified vulnerabilities but also to build resilience and foster a more secure and reliable pharmaceutical ecosystem. Understanding these dynamics is crucial, as highlighted in the trend report of the risk of medicine field in singapore, which underscores the urgent need for a comprehensive and adaptive approach to risk mitigation.
-
Strengthening Regulatory Oversight and Enforcement Efforts
A cornerstone of effective risk management in the medicine sector is a robust and adaptive regulatory framework, backed by diligent enforcement. Singapore’s Health Sciences Authority (HSA) plays a pivotal role in this regard, ensuring the safety, quality, and efficacy of health products. However, continuous strengthening is essential. This involves updating regulations to address emerging threats like sophisticated counterfeit medicines, ensuring the integrity of the pharmaceutical supply chain, and adapting to new medical technologies. Enhanced post-market surveillance, including comprehensive pharmacovigilance systems, allows for the early detection and assessment of adverse drug reactions or quality defects once products are in use. Furthermore, strengthening inspection capabilities and implementing stricter penalties for non-compliance act as powerful deterrents against illicit activities. Organizations must prioritize stringent adherence to Good Manufacturing Practices (GMP) and Good Distribution Practices (GDP), investing in internal compliance teams and regular audits. This commitment to regulatory compliance not only mitigates risks but also reinforces Singapore’s reputation as a trusted hub for quality healthcare products.
-
Adopting Advanced Risk Assessment and Monitoring Technologies
The digital revolution offers unparalleled opportunities for enhancing risk management in the medicine sector. Advanced technologies such as Artificial Intelligence (AI), Machine Learning (ML), and blockchain are transforming how risks are identified, assessed, and monitored. Predictive analytics can forecast potential supply chain disruptions, allowing companies to pre-empt shortages or overstocking. Real-time tracking and tracing systems, often powered by blockchain, enhance supply chain visibility, making it easier to pinpoint the origin of counterfeit products or quality deviations. Furthermore, AI-driven tools can augment pharmacovigilance by analyzing vast amounts of data from clinical trials, patient registries, and social media to detect subtle patterns of adverse drug events more quickly than traditional methods. Integrating these technologies into a comprehensive digital health strategy enables organizations to move from reactive problem-solving to proactive risk mitigation. Investing in robust cybersecurity measures is equally critical, protecting sensitive patient data and proprietary manufacturing information from increasingly sophisticated cyber threats. This technological pivot is vital for maintaining resilience and ensuring patient safety in an interconnected world.
-
Enhancing Collaboration Across the Healthcare Ecosystem
No single entity can effectively manage the multifaceted risks within the medicine sector alone. A collaborative, ecosystem-wide approach is paramount. This involves fostering strong partnerships among government bodies (like HSA, Ministry of Health), pharmaceutical manufacturers, distributors, healthcare providers, research institutions, and even patient advocacy groups. Information sharing platforms can facilitate the rapid dissemination of alerts regarding product recalls, emerging safety concerns, or supply chain vulnerabilities. Joint training programs and simulated exercises can prepare all stakeholders for coordinated responses to crises, such as pandemics or large-scale product contaminations. Public-private partnerships can drive innovation in risk mitigation strategies, accelerate the development of new safety standards, and co-invest in critical infrastructure, such as secure cold chain logistics or advanced laboratory testing capabilities. By breaking down silos and promoting open communication, Singapore can build a more integrated and resilient healthcare ecosystem, capable of responding swiftly and effectively to any challenge, ultimately safeguarding the continuous availability and safety of essential medicines for its population.
Future Outlook and Recommendations for 2026 and Beyond
As the healthcare landscape in Singapore continues its rapid evolution, driven by technological advancements, demographic shifts, and emerging health threats, the imperative to manage and mitigate risks in the medicine field becomes ever more critical. This forward-looking perspective, building upon insights from recent trends, outlines strategic recommendations for stakeholders to navigate the evolving risk landscape effectively in the coming years and ensure robust patient safety. By proactively addressing potential challenges and embracing innovation, Singapore can reinforce its position as a leader in safe and effective healthcare delivery. The recommendations below are designed to foster resilience and adaptability within the system, preparing it for the complexities of 2026 and beyond.

1. Proactive Policy Adjustments for Future Healthcare Challenges
Singapore’s regulatory framework has historically been responsive and robust, yet the pace of change demands an even more proactive stance. For 2026 and beyond, policymakers must anticipate future healthcare challenges, such as the increasing prevalence of multi-drug resistant organisms, the rise of personalized medicine, and the integration of AI and big data in diagnostics and treatment. Recommendations include establishing a dynamic regulatory sandbox for novel medical technologies, allowing for controlled testing and rapid policy adaptation. This approach facilitates innovation while maintaining stringent safety standards. Furthermore, policies should focus on enhancing cross-border collaboration for pharmacovigilance, especially with the globalized supply chain of medicines. Developing agile guidelines for the use of genomics in prescribing and managing drug interactions will also be crucial. Regular reviews of existing legislation, perhaps on a biennial basis, will ensure that policies remain pertinent and effective in safeguarding public health against emerging risks.
2. Investment in Research & Development for Risk Prevention
Sustained investment in research and development (R&D) is paramount to staying ahead of potential risks in the medicine field. This includes funding for basic science research to understand disease mechanisms better, as well as translational research focused on developing new diagnostics, therapeutics, and preventative strategies. Prioritizing R&D in areas like antimicrobial resistance, advanced drug delivery systems, and real-time monitoring of drug efficacy and safety will yield significant returns. For instance, developing AI-driven systems capable of predicting adverse drug reactions based on patient profiles and genomic data could revolutionize patient safety. Collaboration between academic institutions, pharmaceutical companies, and government agencies should be incentivized through grants and partnerships. An external authoritative source highlighting the importance of such collaboration in health security is the World Health Organization (WHO), which consistently advocates for global investment in health R&D, particularly in areas like pandemic preparedness and antimicrobial resistance, as detailed in their ongoing initiatives and reports on global health priorities. For example, the WHO’s work on Antimicrobial Resistance underscores the urgent need for R&D to combat this growing threat, directly impacting the safety and efficacy of medicines worldwide. Enhancing local manufacturing capabilities for essential medicines and vaccines, supported by R&D, will also mitigate supply chain risks and ensure national resilience.
3. Fostering a Culture of Safety, Transparency, and Innovation
Beyond policy and R&D, cultivating an organizational culture that prioritizes safety, transparency, and continuous innovation is fundamental. Healthcare institutions should implement robust incident reporting systems that encourage non-punitive reporting, allowing for systemic learning from errors rather than individual blame. This fosters an environment where healthcare professionals feel safe to report near misses and adverse events, leading to a deeper understanding of underlying systemic vulnerabilities. Transparency with patients about potential risks and benefits of treatments, coupled with shared decision-making models, empowers individuals and builds trust within the healthcare system. Regular training programs focused on medication safety, risk communication, and the ethical implications of new technologies are essential for all healthcare staff. Moreover, fostering an innovative mindset means encouraging pilot projects for new safety protocols, leveraging digital health solutions for medication management, and continuously evaluating their effectiveness. Singapore’s commitment to patient-centric care is already strong, and further embedding these principles will ensure that the future of medicine is not only advanced but also inherently safe and trustworthy. This comprehensive approach aligns with the findings often highlighted in the trend report of the risk of medicine field in Singapore, emphasizing continuous improvement and proactive measures in managing medication-related risks.
Partner with Shelby Global
You are looking for reliable HR Sevice Suppliers? Contact Shelby Global Now! To connect with verified talents and upgrade your orginization.
—————————————
References
– :
– Antimicrobial Resistance – World Health Organization: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
– :
– HSA Regulatory Guidances: https://www.hsa.gov.sg/health-products/regulatory-guidances
– WHO’s work on Antimicrobial Resistance: https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance




























































































































![TOP 4 recruitment agency in vietnam 2026 125 TOP 4 recruitment agency in vietnam 2026 [highlight Shelby Global]](https://shelbyglobal.com.vn/wp-content/uploads/2026/04/TOP-4-recruitment-agency-in-vietnam-2026-highlight-Shelby-Global-1.webp)